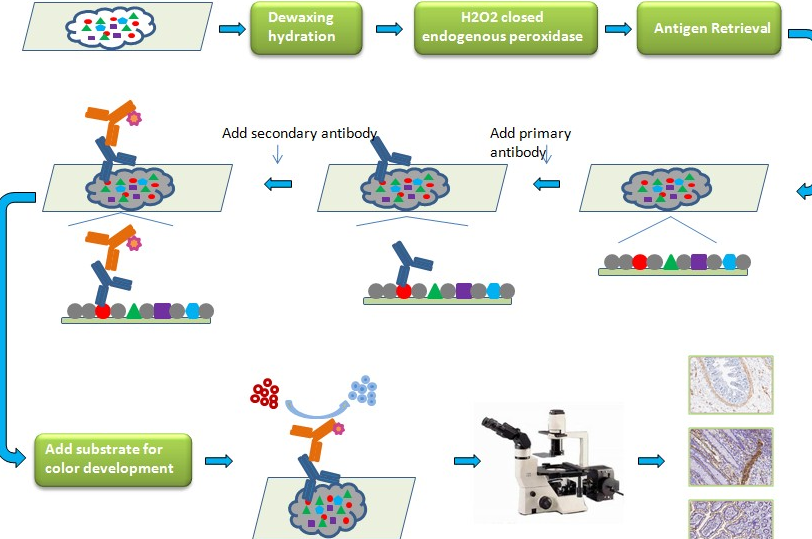
Immunohistochemistry Protocol – Avidin / Biotin Complex (ABC) Method
The principle of the ABC detection kits is based on the affinity of avidin (or streptavidin) for biotin. In fact, the abbreviation ABC stands for Avidin / Biotin Complex.
In general, ABC detection kits are composed of:
- A biotinylated secondary antibody
- From an Avidin / Enzyme complex
Kits may or may not contain blocking and dilution buffers and substrate depending on the formats.
I. Materials required
Reagents
- One (or more) primary antibody (s) coupled or not and specific to your molecule (s) of interest
- Revelation kit
- Chromogenic
Buffers
- Xylene
- 70%, 80% and 95% alcohol baths
- Hydrogen peroxide (H2O2)
- Tris EDTA, Tris HCl
- Tween-20
- Hematoxylin
- Mounting medium
Equipment
- Pipettes and a Pipet-aid
- Water bath
II. Duration of the experiment
- 10 min of peroxidase blocking
- 30 min of antigen unmasking (if necessary)
- 30-60 min incubation with the primary antibody
- 10 min of incubation with the biotinylated secondary antibody
- 10 min incubation with the streptavidin / Peroxidase conjugate
- 5 min of chromogen
- Hematoxylin / Eosin staining (optional)
- TOTAL: 2-3 hours
III. Procedure
- Endogenous peroxidase blocking: Apply 2 drops (100 µl), or a volume sufficient to cover the tissue section, of endogenous peroxidase blocking reagent (3% H2O2 solution) and incubate for a few minutes. Rinse the slide with distilled water.
- Unmasking of the antigen (HIER treatment) depending on the primary antibody used: Unmasking as indicated on the technical sheet of the primary antibody then rinsing 3 x 2 min with PBS.
- Pre-blocking: Apply 2 drops (100 µl), or a volume sufficient to cover the section with tissue, pre-blocking solution and incubate 10 min. Remove the solution but do not rinse.
- Primary antibody: Users should optimize the dilution and incubation time according to the antibody used. Apply 2 drops (100 µl), or a volume sufficient to cover the tissue section, with primary antibody and incubate 30-60 min. Rinse with PBS 3 x 2 min.
- Secondary antibody: Apply 2 drops (100 µl), or a volume sufficient to cover the tissue section, with biotinylated secondary antibody and incubate for 10 min. Rinse 3 x 2 min with PBS
- Streptavidin-HRP Conjugate: Apply 2 drops (100 µl), or a volume sufficient to cover the tissue section, of Streptavidin-HRP conjugate and incubate for 10 min. Rinse 3 x 2 min with PBS
- Substrate / Chromogen: Add 1 drop or 2 drops (for better sensitivity and contrast) of concentrated chromogen to 1 ml of substrate. Shake well, protect from light and use within 5 hours. Apply 2 drops (100 µl), or a volume sufficient to cover the tissue section, with chromogenic solution and incubate for 5 minutes. Rinse 3 x 2 min with distilled water.
- Hematoxylin: Counterstain the sample with 2 drops (100 µl), or a volume sufficient to cover the tissue section, and incubate for 10-20 sec. Rinse thoroughly for 1-2 min with distilled water. Place slides in PBS until a blue color is obtained (30-60 sec).
- Assembly: Follow the technical data sheet for the assembly liquid.
Notes:
- The fixation, the thickness of the tissue sections, the unmasking of the antigen as well as the dilutions and incubation time of the primary antibody can significantly affect the results. Users should take all factors into consideration and determine the optimal conditions.
- The coloration of the tissue is dependent on the treatments carried out on the tissues before the immunohistochemistry. Poor tissue preparation can lead to false negatives and widely divergent results from experiment to experiment.
- If you are using kits containing all reagents for IHC, these are tested together to ensure the best possible results. It is therefore advisable not to mix reagents from different lots.
- Do not allow tissue samples to dry for the duration of the protocol and use a humid chamber for incubations.